A meeting to decide which African country will be the host country for the African Medicines Agency (AMA) is underway and at the last count, expressions of interest in hosting the agency have been received from Uganda, Algeria, Egypt, Morocco, and Rwanda, Tanzania, Tunisia and Zimbabwe.
The decision is expected to be one of the outcomes at the ongoing first Ordinary session of the Conference of State Parties (CoSP) of the AMA, in Addis Ababa, Ethiopia from 1 to 3 June 2022.
The CoSP is reviewing the AMA assessment report and at the end of their meeting, it will make recommendations on the host country of the AMA Headquarters. A final decision on this recommendation will be made at the next assembly of the African Union.
Uganda is one of the countries vying to host the AMA Headquarters and the country’s delegation to the CoSP meeting is led by the country’s Minister of Health, Dr Jane Aceng, and Dr Diana Atwine, a Permanent Secretary in the Ministry of Health.
Zimbabwe’s team is a four-member delegation led by the country’s Health and Child Care Deputy Minister, Dr John Mangwiro.
Sierra Leone is also attending the session led by Minister of Health and Sanitation Dr Austin Demby.
In his remark at the official launch of the Africa CDC’s Ministerial Executive Leadership Program (MELP), Dr John Nkengasong, Africa CDC’s outgoing Director General confirmed the CoSP’s outcome would play a critical role in deciding the future of the agency.
“The launch of the AMA is really another landmark initiative of the African Union as part of the Agenda 63. I can’t wait to hear the outcome of the deliberations. It’s going to be transformational when the AMA and we are truly excited with that,” Nkengasong said.
The three-day ministerial meeting will deliberate on the priorities and next steps to establish the AMA.
From Geneva to Addis Ababa
At last week’s World Health Assembly, Amref Health Africa’s Desta Lakew, on behalf of the African Medicines Agency Treaty Alliance (AMATA) described AMA as an initiative of the African Union aimed at strengthening the regulatory environment to guarantee access to quality, safe and efficacious medicines, medical products, and technologies on the continent.
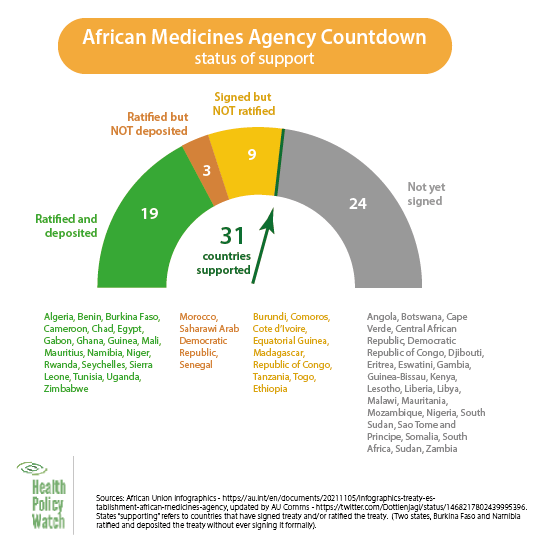
Countries that have signed the treaty, according to AMATA, are Algeria, Benin, Chad, Ghana, Madagascar, Mali, Morocco, Rwanda, Saharawi Arab Democratic Republic, Senegal, and Tunisia. But about four years after the AU Assembly adopted the AMA Treaty, Lakew noted that some Member States still haven’t given it the authority to fulfil its mandate.
“The increased risk of disease outbreaks in the region and globally has made access to medical products a priority. Trade-in locally produced medical products, including Personal Protective Equipment, across African countries will be hampered if we do not have a harmonized regulatory environment,” Lakew told WHA 75.
At the CoSP meeting, the AU called on all member states to sign the Treaty to see improved regulation and easy flow of medical products and technologies, which are expected to be reechoed. Lakew described the call as an urgent matter “for the sake of ensuring that there is equitable access to quality, safe, efficacious medicines and technologies for a healthy Africa”.
Last month, Health Policy Watch reported Kenya was preparing to join the AMA alongside thirty-one other African Union member states following the Kenyan cabinet’s decision to approve the ratification of the African Union Treaty.
Article first published on https://healthpolicy-watch.news/eight-countries-compete-to-host-african-medicines-agencies-at-crucial-au-meeting/
